Christina Nunez is contract writer for Argonne National Laboratory. Nunez can be reached at [email protected].
Humans have been filtering liquids for centuries. But as anyone in the water industry knows, the science of clean water is anything but static. New technologies continue to push the boundaries of what membranes can achieve in separating liquids from contaminants.
At the U.S. Department of Energy’s (DOE) Argonne National Laboratory, researchers are developing these advances, inspired by everything from the cutting edge of nanoscience to the oldest of writing materials. Membrane research is an essential part of a broader mission at Argonne to improve society’s ability to treat and reuse water.
Most commercial membranes used to clean liquids rely on a small set of polymers. These materials have their advantages, but the current chemistry is limited, contributing to common pitfalls such as fouling and degradation.
“A lot of our work involves manipulating the interface—the surface of where the material encounters the fluid,” said Seth Darling, director of the Institute for Molecular Engineering and the Advanced Materials for Energy-Water Systems Energy Frontier Research Center at Argonne.
Fending Off Fouling
This advanced interface engineering is creating new possibilities for membranes. One invention involves a multifunctional membrane that, when activated by light, can clean itself and degrade pollutants. The main ingredient driving this pollutant-fighting power is a coating of titanium dioxide.
Though titanium dioxide’s ability to accelerate chemical reactions under ultraviolet (UV) light is well established, Argonne researchers employed it with two twists. First, they added small amounts of nitrogen to the titanium dioxide, “doping” it so that visible light, not just UV light, would bring out the photocatalytic properties.
Second, researchers used atomic layer deposition (ALD), a technique for creating thin films often used in the semiconductor industry, to place the coating on the membrane. Unlike the typical method of dipping the membrane into a solution, ALD grows the coating one molecular layer at a time. This allows all of the membrane surfaces, including the internal nanopores, to be coated uniformly and precisely.
The specially coated membrane can degrade both pore-clogging foulants and common water pollutants, including endocrine disruptors and hormones that pose health and environmental concerns.
“Almost anything that’s organic [carbon-based] will get broken down by this type of chemistry,” Darling said.
Novel Membrane Techniques
As one of the leading research institutions with expertise in ALD, Argonne has been positioned to pioneer its use in membrane fabrication.
“What’s nice about this technique is that the resulting coating is extremely robust,” Darling said. “It’s tightly bound to the membrane itself and basically will never come off, unlike those deposited with conventional strategies.”
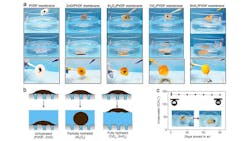
ALD also was used to create an improved Janus membrane, a two-faced gateway that could help optimize or enable a range of industrial processes from treating wastewater to making biofuels. Traditional means of creating Janus membranes rely on processes that can be difficult to control.
Researchers deposited a water-attracting layer of aluminum oxide onto a water-repellent polypropylene membrane, creating a stable Janus membrane that could be used in fine-bubble aeration, conveying tiny gas bubbles into liquid. In this instance, the use of ALD improved the coating’s stability and controlled how deeply it penetrated the one side.
The ability to make Janus membranes with this level of precision and stability could introduce new levels of efficiency in a variety of industrial processes. At wastewater treatment plants, where aeration is used to help break down contaminants, optimizing the bubbling process this way could reduce energy expenditures.
Improved Janus membranes also could accelerate emulsification or demulsification of oil-water mixtures, both important in a variety of manufacturing processes.
Argonne also is developing coatings designed to address this specific problem of oil separation. A metal oxide coating placed on off-the-shelf commercial polymer membranes, for example, produces thin films of water-loving, oil-repelling materials on the membrane surface.
“The coating is just a few nanometers in thickness. If the coating were thicker than this, it would close off the tiny pores,” Darling said. “What you want is a minimal change of the pore structure, but you want to change the chemistry of the substance lining those pores.”
Researchers tested different metal oxides to see which ones worked best, finding that tin oxide and titanium oxide formed the tightest bonds with water molecules, capturing them and layering them across the surface.
“Just from my knowledge of the oil and gas sector, if we could make a membrane that performs to even a fraction of what we’ve seen in lab testing, it will be a phenomenal improvement over what’s available now. That represents a huge savings,” said Aaron Fluitt, Argonne’s business development executive handling the technology.
Creatively Lowering Costs
Some breakthroughs at Argonne balance advanced technology with bursts of creativity. While searching for affordable photothermal materials that could be used in steam purification of water, Chinese-born researcher Hao-Cheng Yang was inspired to experiment with an ancient ink he encountered in childhood as a coating.
In solar steam generation, a light-absorbing material concentrates heat from the sun to evaporate water at the surface, leaving salt and contaminants behind. Researchers were looking for dark liquids that could accelerate solar heating. Chinese ink, a traditional writing material made with soot, glue and a preservative, turned out to have the perfect combination of qualities.
Unlike the ink in modern pens, Chinese ink is designed to have staying power on surfaces of all kinds, from fabric to pottery. Its viscosity keeps it from penetrating too far down into the substrate, which could be membranes, wood, or another material. However, the carbon-based paint’s biggest asset may be its propensity to absorb light across the visible spectrum and into the infrared range, where most of the sun’s energy lies.
“This is a very effective material at absorbing that broad range of light,” Darling said., “which makes it fantastic for this application of solar steam generation.”
Since the ink is water-soluble, the researchers used ALD to seal it with a transparent coating of titanium dioxide, protecting it from dispersing into the surrounding water. Tested under simulated sunlight on various substrates, including membranes, the combination proved to be a stable, potent steam generator.
Another creative application of technology at Argonne involves a new interface engineering technique called sequential infiltration synthesis (SIS). Similar to ALD, the method can be used to alter a membrane’s surface using chemical vapors. But instead of adding layers to the top of a membrane as ALD would do, SIS grows material inside the membrane structure itself, changing its chemistry without significantly affecting the pore shape.
SIS makes use of the space between molecules in membrane polymers, penetrating the surface and diffusing into it with an inorganic material. In their proof of concept, Darling and colleagues used SIS to plant the “seeds” for aluminum oxide and grew it within polyethersulfone (PES) ultrafiltration (UF) membranes, making them more resilient without compromising filtration ability.
The SIS technique enables a range of improvements to membranes: the ability to prevent foulants from attaching to the surface, for example, or resistance to solvents that might be needed in an industrial setting but would dissolve conventional membrane materials.
SIS also was used to create Oleo Sponge, which captures oil from water. In that case, a metal oxide grown within the sponge’s surface serves as a grafting site for oil-loving molecules. A similar strategy could be employed with membranes, where molecules are grafted to lend selectivity or some other desired characteristic.
The research underway at Argonne is conducted with commercial feasibility and costs in mind. In making membranes more robust and multifunctional, these techniques could help eliminate the expense of cleaning or replacing them, while avoiding the labor and operational costs associated with maintenance shutdowns.
Darling is aware that the advances developed with a certain problem in mind could lend themselves to scenarios he and his fellow scientists probably have not even thought of yet.
“One thing I learned from the Oleo Sponge is that you can’t envision all the possible applications at the beginning,” Darling said. “We anticipated interest from oil companies, but we’ve also heard from the cosmetics industry and from sporting goods manufacturers.”
Industry will play a key role in advancing these technologies, either by collaborating with Argonne to further develop them or by licensing them outright.